Nuvaxovid
Nuvaxovid dispersion for injection. On December 20 2021 the.

Novavax S Vaccine Nuvaxovid Vaktsineeri Ee
88 experienced pain.

. This vaccine is currently being used in Sweden and as of date a total of 7000. Folkhälsomyndigheten rekommenderar att det proteinbaserade covid-19-vaccinet Nuvaxovid inte ges till personer som är 30 år och yngre. 21 hours agoThe US company Novavax came up with another vaccine to fight the virus - Nuvaxovid.
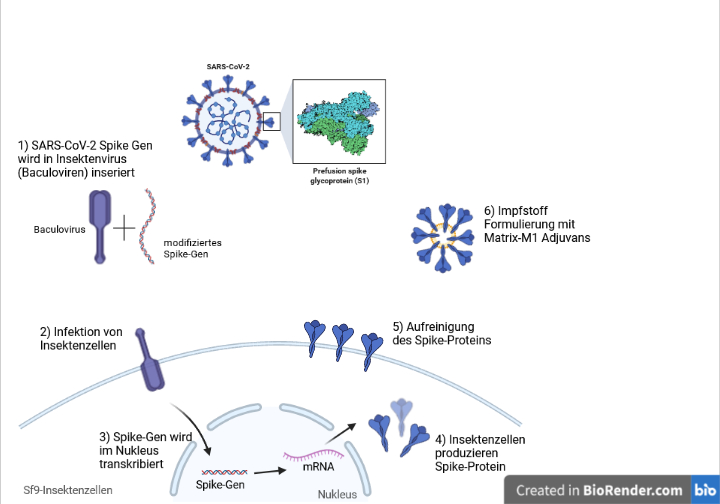
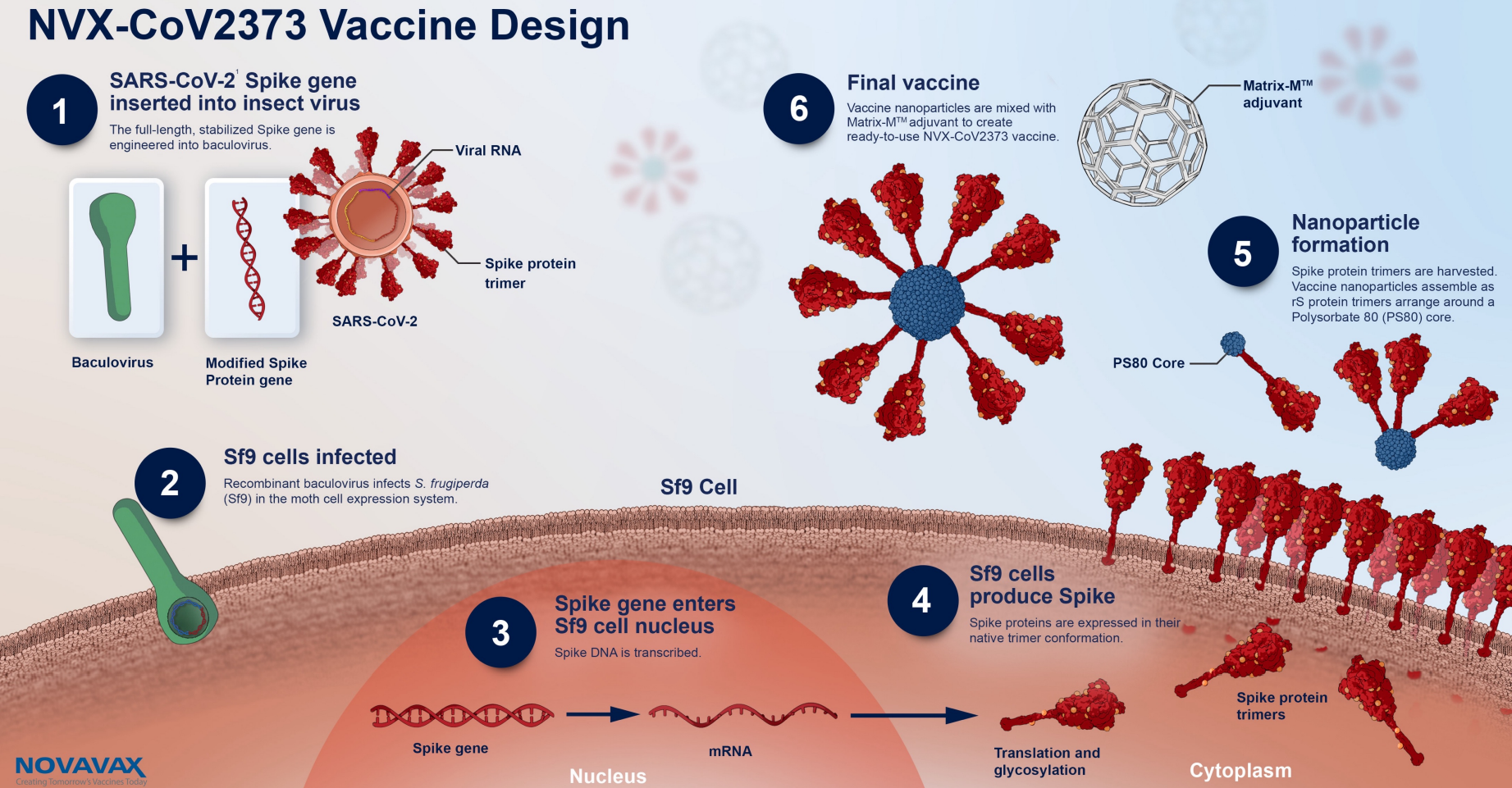
About Nuvaxovid NVX-CoV2373 Nuvaxovid is a protein-based vaccine engineered from the genetic sequence of the first strain of SARS-CoV-2 the virus that causes. Nu stoppar Folkhälsomyndigheten användningen bland personer som är 30. Rokotteesta ei myöskään ole haittaa vaikka.
After the approval of the mRNA vaccines Corminaty BiontechPfizer Spikevax Moderna and the vector-based vaccines Vaxzevria Astra Zeneca and Covid-19 Vaccine Janssen a further. Qualitative and quantitative composition. 1 day agoSverige Covid-19-vaccinet Nuvaxovid skulle erbjudas till personer som var tveksamma till vaccinationen.
Nuvaxovid is composed of purified full-length SARS-CoV-2 recombinant spike S protein that is stabilised in its prefusion conformation. This is a multidose vial. Esimerkiksi aiemmin sairastettu koronavirustauti ei estä rokotuksen antamista.
About 14m doses of the Nuvaxovid vaccine developed by the US biotech company Novavax are to arrive in Germany this week the countrys health minister Karl Lauterbach. COVID-19 Vaccine recombinant adjuvanted 2. The agency said that younger people who had recently been vaccinated with Nuvaxovid had no.
Information about the COVID-19 vaccine Nuvaxovid approved by the MHRA on 03 February 2022. Det eftersom att data från. The Summary of Product Characteristics is a description of a.
The Nuvaxovid vaccine a protein-based vaccine engineered from the genetic sequence of the first strain of the SARS-CoV-2 virus which causes COVID-19. Nuvaxovid offers a high level of protection against COVID-19 which is a critical need in the current pandemic. Like the Novavax vaccine side effects were more.
1 day agoAround 7000 doses of Nuvaxovid have already been administered in Sweden. Data från Australien pekar mot en ökad. Nuvaxovid-rokote sopii lähes kaikille aikuisille.
Det proteinbaserade covid-19-vaccinet Nuvaxovid inte ska ges till personer som är 30 och yngre meddelar Folkhälsomyndigheten. The Technical Advisory Group for Emergency Use Listing listed Nuvaxovid NVX-CoV2373 vaccine against COVID-19 and Covovax NVX-CoV2373 vaccine against COVID-19. The first batch of Nuvaxovid is expected to arrive in.
First Approval of the Protein-Based Adjuvanted Nuvaxovid NVX-CoV2373 Novavax Vaccine for SARS-CoV-2 Could Increase Vaccine Uptake and Provide Immune. Clinical trials showed that the vaccine has around 90 efficacy. Nuvaxovid contains a version of a protein found on the.
Nuvaxovid COVID-19 vaccines are available for use in the United Kingdom as of September 27 2022. Nuvaxovid is composed of purified full length severe acute respiratory syndrome coronavirus 2 SARS-CoV-2 recombinant spike S protein that is stabilised in its prefusion conformation. This will enable us to start offering the Nuvaxovid.
1 day agoBakgrunden till beslutet är signaler om ökad risk för hjärtmuskelinflammation myokardit och hjärtsäcksinflammation perikardit. 16 fever including 14 severe cases. Beslutet är temporärt och gäller från.
Nuvaxovid is the first protein-based COVID-19 vaccine granted. Name of the medicinal product. 1 day agoPublicerad idag 0702.
The Nuvaxovid NVX-CoV2373 Novavax vaccine is a recombinant spike S protein nanoparticle vaccine combined with the Matrix-M adjuvant. The addition of the saponin-based.

Cdc Endorses Novavax Covid Shot For Adults Fortune

Novavax Stock Looks Like A Good Value With Its New Combined Vaccine

Informacoes Sobre A Vacina Nuvaxovid Novavax Contra Covid 19 Australian Government Department Of Health And Aged Care

Covid Vaccine Novavax Requests Who To Expand Emergency Use Listing Of Nuvaxovid For Adolescents Aged 12

Novavax Eyeing The Covid Vaccine Hesitant And Kids Unveils New Education Campaigns As Nuvaxovid Nears Us Finish Line Fierce Pharma
Nuvaxovid Otazky A Odpovedi K Vakcine Od Spolecnosti Novavax Kurzy Cz
Fda Authorizes Novavax Covid 19 Vaccine For Emergency Use In Us Abc News

Novavax Covid Vaccine Nuvaxovid Medcity News

Vaccine In Short For Nuvaxovid Novavax Publication Medicines Evaluation Board
Ema Chmp Recommends Cma For Novavax S Covid 19 Vaccine As Booster

Novavax Covid 19 Vaccine Nuvaxovid Data On Side Effects

Middlesex London Health Unit Are You Interested In The Novavax Nuvaxovid Covid 19 Vaccine Mlhu Is Receiving A Limited Supply Of The Vaccine This Spring Call 1 226 289 3560 To Be Added To The Waitlist

Canadian Trademarks Details Nuvaxovid 2163962 Canadian Trademarks Database Intellectual Property And Copyright Canadian Intellectual Property Office Innovation Science And Economic Development Canada

Ema Recommends Nuvaxovid For Authorisation In The Eu Certifico Srl
Novavax Nuvaxovid Covid 19 Vaccine Approved In South Korea For Use In Adolescents Aged 12 Through 17 World Pharma Today
Novavax S Covid 19 Vaccine Moves Closer To Fda Authorization Decision Wsj

Ministry Of Health Singapore On Instagram Registration For The Nuvaxovid Vaccine By Novavax Has Begun Individuals Aged 18 Years And Above May Receive The Vaccine For Their Primary